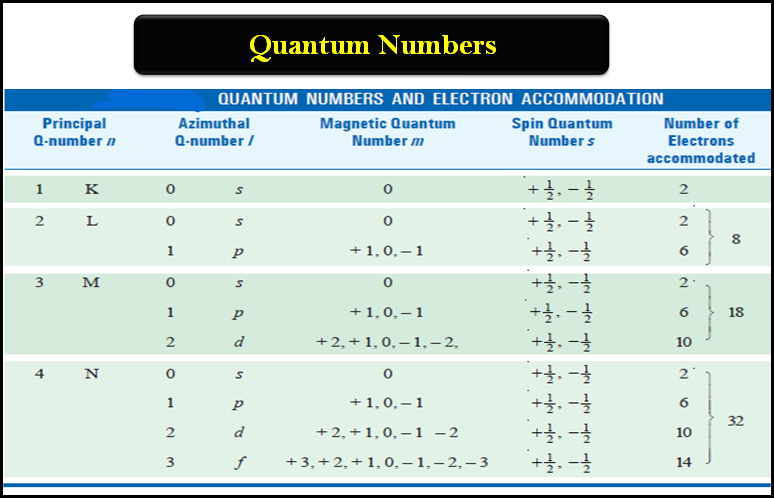
As n increases, the electron orbital becomes larger and the electron spends more time farther from the nucleus.Ĭlearly the three quantum numbers, n, ℓ, m ℓ, are not enough to completely describe the state of the H-atom, Another quantum number is required to describe whether it goes up or down in a spatially varying magnetic field. The principal quantum number has integral values of n = 1, 2, 3. For an electron trapped by its electrostatic attraction to positively charged nucleus we use the three following quantum numbers to describe the electron state (orbital). Let's look at the quantum numbers needed to label the possible standing waves or states of an electron trapped by its electrostatic attraction to a positively charged nucleus. Normally we operate in 4 dimensions (3 space and 1 time dimension), so in general we will need 4 quantum numbers to fully specify an electron state ( i.e., standing wave). Now it turns out that if you consider an electron trapped inside a sphere (instead of in a one-dimensional box) it will have standing waves that are very similar to an electron bound to a positively charged nucleus by electrostatic attraction. Quantum Numbers - Specifying the electron state
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
